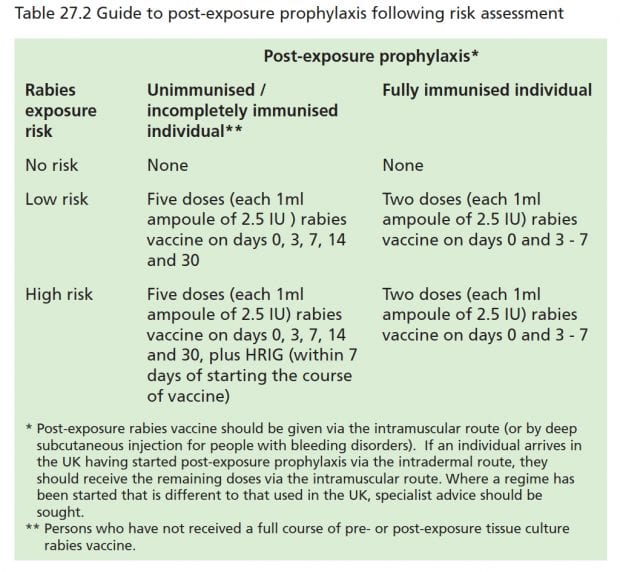
Intradermal post-exposure rabies vaccination with purified Vero cell rabies vaccine: Comparison of a one-week, 4-site regimen versus updated Thai Red Cross regimen in a randomized non-inferiority trial in the Philippines - ScienceDirect

Viruses | Free Full-Text | Factors Associated with Dog Rabies Immunization in Changsha, China: Results of a Cross-Sectional Cluster Survey, 2015–2021

Safety and immunogenicity of a mRNA rabies vaccine in healthy adults: an open-label, non-randomised, prospective, first-in-human phase 1 clinical trial - The Lancet

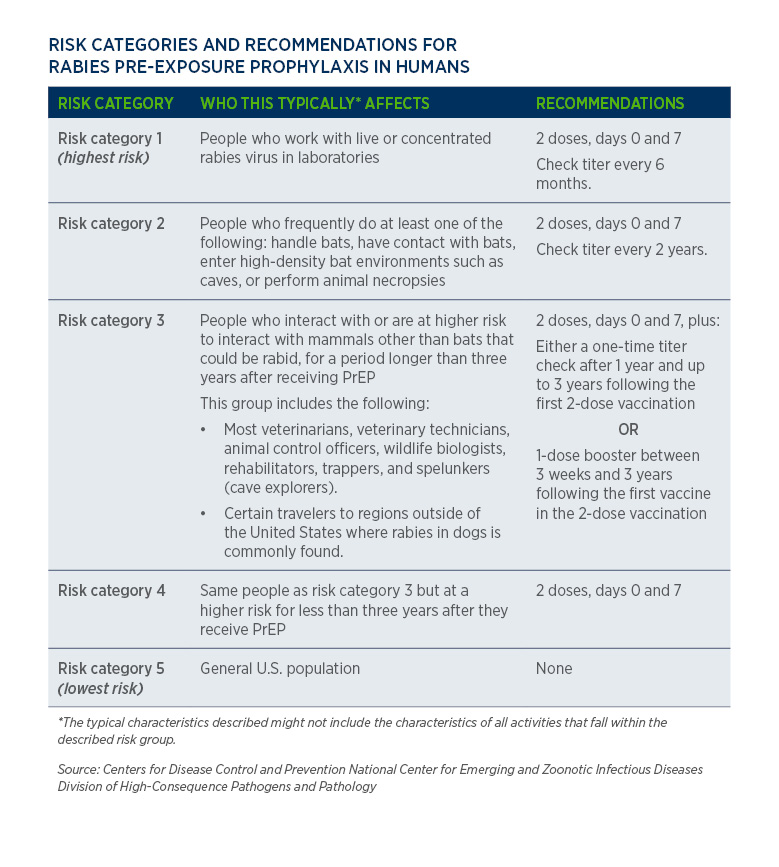
Use of a Modified Preexposure Prophylaxis Vaccination Schedule to Prevent Human Rabies: Recommendations of the Advisory Committee on Immunization Practices — United States, 2022 | MMWR



:strip_icc()/how-long-are-rabies-shots-good-3385625_FINAL-b3b90b78675e4a9ca4102b405fecf74c.jpg)


![PDF] The Route of Administration of Rabies Vaccines: Comparing the Data | Semantic Scholar PDF] The Route of Administration of Rabies Vaccines: Comparing the Data | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/05edbf7c0547fddc89416fbcc67a2664c879ff79/3-Table1-1.png)

:max_bytes(150000):strip_icc()/rabies-overview-4156466_final-8c26e268da134ff1b2c08160d68f42c4.png)